Authorized Distributor (Domestic and Overseas Partners)
Integration with global markets, fully compliant with regulations
- Providing cross-border agency and regulatory support for pharmaceuticals, APIs, and medical devices
- Meeting GDP documentation requirements and data traceability standards
- Ensuring full compliance, transparency, and complete traceability of information
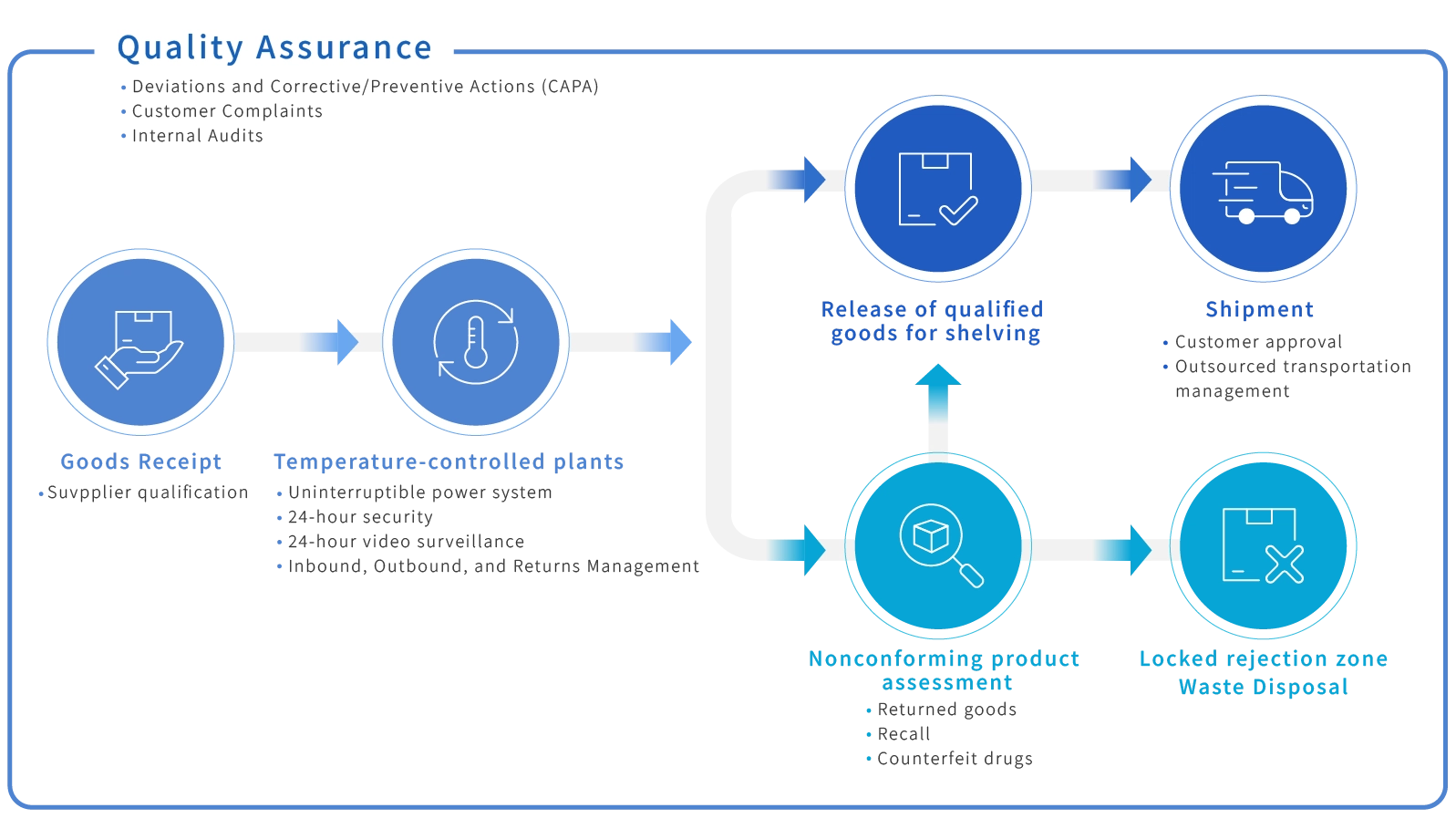
GDP Warehouse Management
Temperature-controlled and standardized storage and transportation processes
- Professional GDP warehouse center with 24-hour environmental monitoring
- Provides ambient storage and distribution
- Detailed records of each shipment’s flow and conditions
- Service strengths | Transparent processes | Quality certifications fully compliant with regulations
Contract Development and Manufacturing (CDMO)
R&D contract support with verifiable data integrity
- Provision of APIs, formulation design, and dosage form development
- Real-time documentation at every stage, compliant with ALCOA+ principles
- Authentic and complete experimental records, ensuring data quality assurance
OEM/ODM Contract Manufacturing
Standardized manufacturing processes with compliant documentation
- Manufacturing environment in accordance with GMP/GDP standards
- Real-time production records with version traceability
- Each batch fully traceable and subject to review
- Controlled documentation and records for packaging and labeling
Regulatory Submission Document Services
Professional regulatory team ensuring compliant document management
- Support for drug registration, IND/NDA applications, and international regulatory requirements
- Document drafting, revision, and review processes fully aligned with GDP standards
- Complete record retention, traceable modifications, and clear version control